Chapter 2: Measurement Basics
2.4. Temperature Units
Learning Objectives
By the end of this section, you will be able to:
- Convert temperature between Celsius and Fahrenheit.
Conversion of Temperature Units
We use the word temperature to refer to the hotness or coldness of a substance. One way we measure a change in temperature is to use the fact that most substances expand when their temperature increases and contract when their temperature decreases. The mercury or alcohol in a common glass thermometer changes its volume as the temperature changes. Because the volume of the liquid changes more than the volume of the glass, we can see the liquid expand when it gets warmer and contract when it gets cooler.
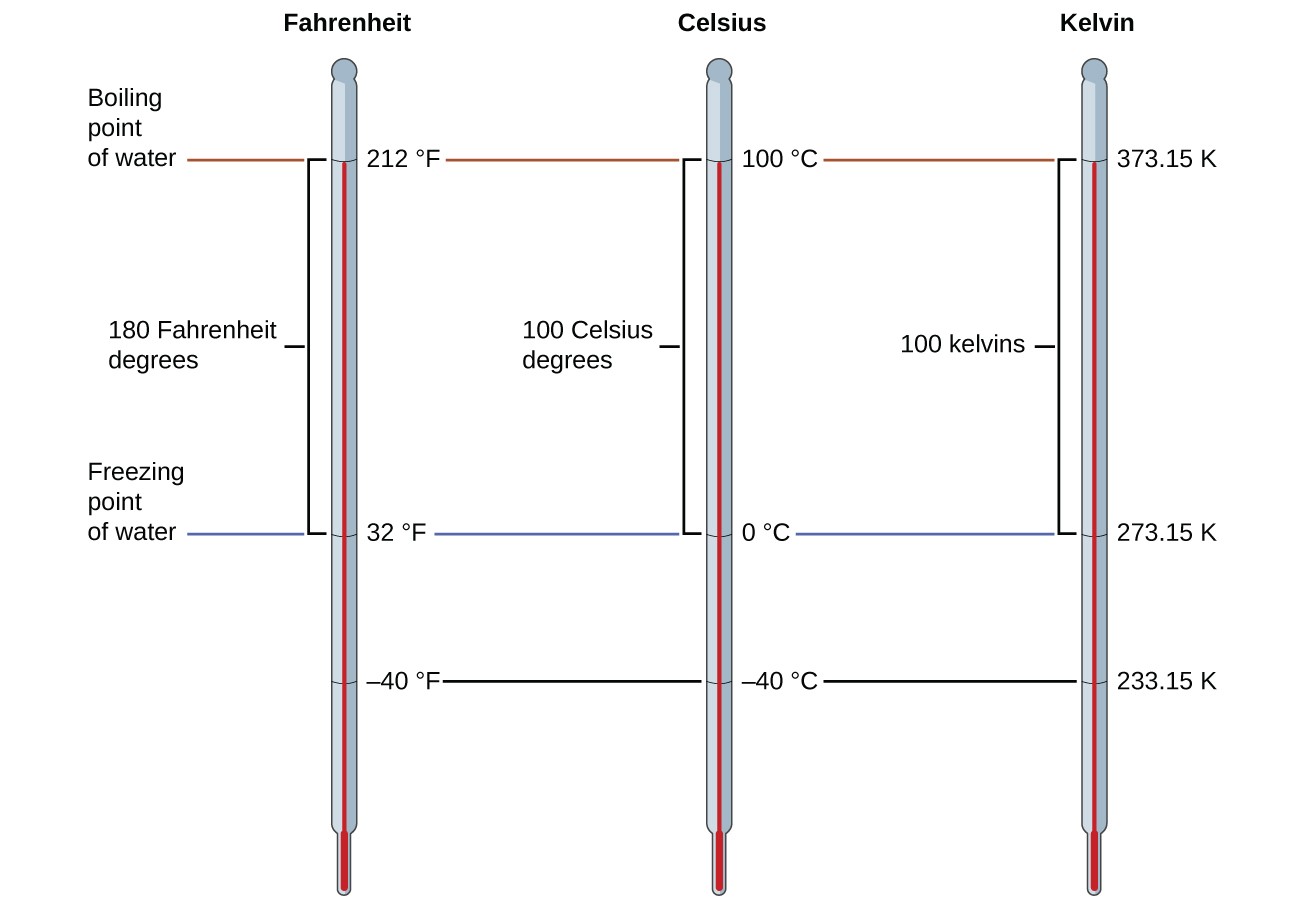
To mark a scale on a thermometer, we need a set of reference values: Two of the most commonly used are the freezing and boiling temperatures of water at a specified atmospheric pressure. On the Celsius scale, 0 °C is defined as the freezing temperature of water and 100 °C as the boiling temperature of water. The space between the two temperatures is divided into 100 equal intervals, which we call degrees. On the Fahrenheit scale, the freezing point of water is defined as 32 °F and the boiling temperature as 212 °F.
An equation for the conversion of temperature from Celsius to Fahrenheit is:
[latex]{T}_{\text{ }^{\circ}\text{F}}=\frac{9}{5}\times{T}_{^{\circ}\text{ C}}+32[/latex]
Rearrangement of this equation yields the form useful for converting from Fahrenheit to Celsius:
[latex]{T}_{\text{ }^{\circ}\text{C}}=\frac{5}{9}\left({T}_{^{\circ}\text{ F}}-32\right)[/latex]
As mentioned earlier in this chapter, the SI unit of temperature is the kelvin (K). Unlike the Celsius and Fahrenheit scales, the kelvin scale is an absolute temperature scale in which 0 (zero) K corresponds to the lowest temperature that can theoretically be achieved. The early 19th-century discovery of the relationship between a gas’s volume and temperature suggested that the volume of a gas would be zero at −273.15 °C. In 1848, British physicist William Thompson, who later adopted the title of Lord Kelvin, proposed an absolute temperature scale based on this concept (further treatment of this topic is provided in this text’s chapter on gases).
The freezing temperature of water on this scale is 273.15 K and its boiling temperature 373.15 K. Notice the numerical difference in these two reference temperatures is 100, the same as for the Celsius scale, and so the linear relation between these two temperature scales will exhibit a slope of [latex]1\frac{\text{K}}{^{\circ}\text{C}}[/latex] . Following the same approach, the equations for converting between the kelvin and Celsius temperature scales are derived to be:
[latex]{T}_{\text{K}}={T}_{^{\circ}\text{C}}+\text{273.15}[/latex]
[latex]{T}_{\text{ }^{\circ}\text{C}}={T}_{\text{K}}-\text{273.15}[/latex]
The 273.15 in these equations has been determined experimentally, so it is not exact. Figure 2.4.1. shows the relationship among the three temperature scales. Recall that we do not use the degree sign with temperatures on the kelvin scale.
Although the kelvin (absolute) temperature scale is the official SI temperature scale, Celsius is commonly used in many scientific contexts and is the scale of choice for non-science contexts in almost all areas of the world. Very few countries (the U.S. and its territories, the Bahamas, Belize, Cayman Islands, and Palau) still use Fahrenheit for weather, medicine, and cooking.
License and attributions:
- Chemistry: Atoms first, Second edition, 2019, Flowers, P. et al. License: CC BY 4.0. Located at https://openstax.org/books/chemistry-atoms-first-2e/pages/1-6-mathematical-treatment-of-measurement-results
